July 5 (UPI) — The U.S. Food and Drug Administration is clearing the way for expanded use of the DigniCap Cooling System that reduces hair loss after chemotherapy.
Hair loss is one of the most common negative side effects of chemotherapy treatment and is used mostly with solid tumor cancers.
The FDA first approved use of the DigniCap System in 2015 for breast cancer patients, announcing on Monday that the cap is now being recommended for patients with solid tumor cancers in other parts of the body as well.
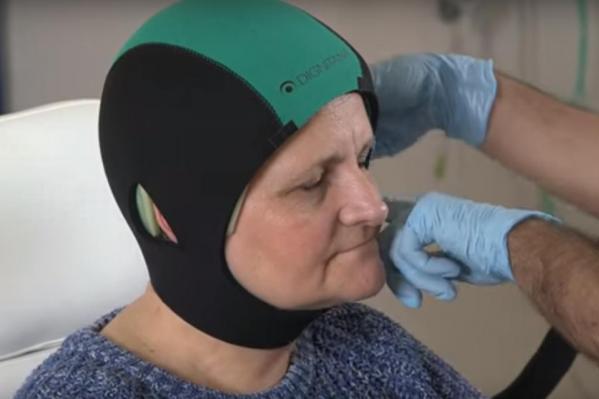
The cooling cap system is a computer-controlled cooling cap that offsets the effects of chemotherapy on hair and minimizes hair loss.
DigniCap had been widely used in other countries before being approved for use in the United States, according to its manufacturer, Dignitana.
“We are pleased to expand the use of this product for cancer patients with solid tumors to potentially minimize chemotherapy-induced hair loss,” Dr. Binita Ashar, director of the Division of Surgical Devices, in the FDA’s Center for Devices and Radiological Health, said in a press release. “Managing the side effects of chemotherapy is a critical component to overall health and quality of life.”
The DigniCap is worn by patients during treatment as it circulates liquid to the cap to cool the scalp. The cap is covered by a second cap made of neoprene, a type of rubber that holds the cooling cap in place and provides insulation to prevent loss of cooling.
The cooling effect constricts blood vessels in the scalp to reduce the amount of chemotherapy that reaches the cells in hair follicles. The cold temperatures decrease the activity of hair follicles, slowing cell division and limiting the effects of chemotherapy on them.
In clinical trials in 2015 that included 122 women with Stage I and II breast cancer undergoing chemotherapy, more than 66 percent of patients treated with DigniCap reported losing less than half of their hair.