SILVER SPRING, Md., Dec. 15 (UPI) — Hair loss has long been a common side effect of chemotherapy but women undergoing the treatment for breast cancer may not have to deal with it for much longer.
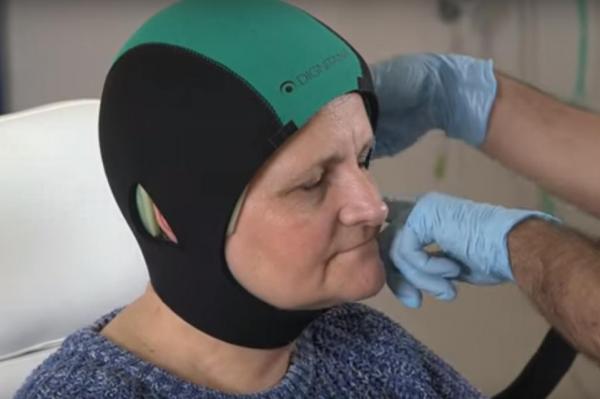
The Food and Drug Administration announced its approval of the DigniCap system, a computer-controlled cooling cap that offsets the effects of chemotherapy on hair and minimizes loss.
DigniCap is used widely outside the United States, according to its manufacturer, Dignitana, and was approved by the agency after a multi-center clinical trial to establish that it works and is safe.
“Some of today’s most powerful, life-saving chemotherapy treatments still cause complete hair loss, a side effect that many women consider to be emotionally devastating,” said Dr. Hope Rugo, director of breast oncology and clinical trials education at the University of California San Francisco, in a press release.
“For the past several years, we’ve worked closely with Dignitana to conduct rigorous clinical trials that demonstrate the safety and effectiveness of the DigniCap system. This FDA clearance means that for many cancer patients in the U.S., chemo-induced hair loss will no longer be a distressing concern.”
The cooling cap is worn during chemotherapy treatment and is controlled by a computer system that circulates cooled liquid intended to constrict blood vessels in the scalp. This decreases the amount of chemotherapy that reaches cells in hair follicles, in addition to the cold temperature lowering the activity of follicles, slowing cell division and reducing further the effects of chemotherapy on hair.
The clinical trial included 122 women with stage 1 or stage 2 breast cancer undergoing chemotherapy, with 7 out of 10 patients keeping at least 50 percent of their hair.
Dignitana also reported patient satisfaction scores were high. On a scale of 0 to 100, participants reported a mean score of 87.5 for satisfaction with the decision to use the cap, 70.9 for hair quantity, and 69.1 for satisfaction with hair quality.
“We are pleased to see a product for breast cancer patients that can minimize chemotherapy-induced hair loss and contribute to the quality of life of these individuals,” said Dr. William Maisel, acting director of the Office of Device Evaluation in the FDA’s Center for Devices and Radiological Health, in a press release. “Managing the side effects of chemotherapy is a critical component to overall health and recovery.”





